Biotech firm wants to grow rice for medicinal supplements
A small company's bid to greatly expand its
genetically engineered rice operation has brought the
international debate over biotechnology home to California
farmers, who largely have avoided the issue directly in recent
years.
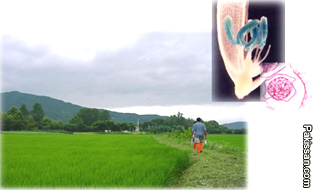 Ventria
Bioscience of Sacramento is pushing hard to gain government
approval to expand its planting of rice engineered with human
DNA to produce medicine in time for this year's growing
season, which is getting underway in a matter of weeks.
Ventria
Bioscience of Sacramento is pushing hard to gain government
approval to expand its planting of rice engineered with human
DNA to produce medicine in time for this year's growing
season, which is getting underway in a matter of weeks.
Within two years, Ventria hopes to have the first marketing
approval to grow rice that produces over-the-counter
treatments for iron deficiencies, diarrhea and other ailments.
The company's timeline could be hampered if it doesn't receive
the necessary state and federal approvals to expand its
operations in the next few weeks. Even the company conceded
U.S. Department of Agriculture approval in time is a long
shot. California regulators also need to approve the plan.
Nonetheless, Ventria's bid to plant more than 100 acres of the
genetically modified rice this year and thousands more in
coming years has touched off widespread disagreement over
biotechnology in the normally insular California rice
industry.
 Though
some California growers already tend to federally approved
fields of genetically engineered cotton and, to a far lesser
extent, modified corn and soy, the state's farmers have in
recent years largely avoided the controversy surrounding
biotechnology's use in the Midwest.
Though
some California growers already tend to federally approved
fields of genetically engineered cotton and, to a far lesser
extent, modified corn and soy, the state's farmers have in
recent years largely avoided the controversy surrounding
biotechnology's use in the Midwest.
Voters in rural Mendocino County last month did vote to ban
biotechnology from its borders. But the ballot measure had
little practical effect, because no biotech crops are grown in
the county. (Related story: Voters in Mendocino ban gen-mod
plants and animals)
Instead, Ventria's application to expand beyond the 100 acres
of experimental rice it has been permitted to grow since 1997
has caused a widespread stir over biotechnology among
California farmers and consumers not seen since the
genetically engineered tomato Flavr Savr was approved for sale
in 1994. The tomato was taken off the market three years later
after it ran into production and shipping problems.
Last week, a sharply divided subcommittee of the California
Rice Commission voted to support Ventria's application to
expand. The application now is pending before the California
Department of Food and Agriculture, which also needs to
approve the company's plans and is expected to rule soon.
 The
6-5 vote came only after months of debate among rice farmers,
many of whom fear losing European and Asian customers
concerned about the biotech rice accidentally mixing with
conventional crops.
The
6-5 vote came only after months of debate among rice farmers,
many of whom fear losing European and Asian customers
concerned about the biotech rice accidentally mixing with
conventional crops.
"We have some wonderful Japanese customers and their concerns
and their confidence is extremely important to us," said Butte
County rice grower Bryce Lundberg. "We should listen to the
Asia market."
Many European and Japanese consumers are mistrustful of
genetically engineered crops and other changes to their food
supply because of government mishandling of mad cow disease in
the past.
Further, some growers in California's $500 million-a-year rice
industry are afraid that a Ventria mistake could harm their
business if biotech rice somehow finds its way into their
conventional crops.
 Previous
biotechnology mistakes like the Starlink controversy in 2000,
when a biotech corn not approved for human consumption was
accidentally mixed with other crops, triggered food recalls
and caused a worldwide drop in corn prices.
Previous
biotechnology mistakes like the Starlink controversy in 2000,
when a biotech corn not approved for human consumption was
accidentally mixed with other crops, triggered food recalls
and caused a worldwide drop in corn prices.
Last year the USDA fined ProdiGene Inc. of College Station,
Texas, some $250,000 for failing to completely remove corn
genetically engineered to produce a pig vaccine before growing
soybeans.
Ventria and the head of the rice commission said there's
little chance of the company's engineered rice mixing with
conventional crops. The company has agreed to grow its rice in
an undisclosed Southern California location, at least 100
miles from the nearest rice farm and 300 miles from the center
of most rice production north of Sacramento.
Nearly all the state's 500,000 acres of rice fields are in
Northern California. Further, rice is "self pollinating" and
the chances of animals, insects and wind mixing biotech
varieties with conventional crops is slim, Ventria's CEO Scott
Deeter said.
"We have multiple safeguards," Deeter said.
Deeter said growing medicines in plants like Ventria intends
could help cut the cost of drug manufacturing.
 Rice
commission chief Tim Johnson said he believes the overseas
markets won't be affected by Ventria's expansion plans. He
said California rice farmers already adhere to strict
guidelines addressing pesticide use that are respected by
international customers and that is not expected to change
because of Ventria's plans.
Rice
commission chief Tim Johnson said he believes the overseas
markets won't be affected by Ventria's expansion plans. He
said California rice farmers already adhere to strict
guidelines addressing pesticide use that are respected by
international customers and that is not expected to change
because of Ventria's plans.
"The concerns are high," Johnson said. "But at the end of the
day, those concerns won't be realized.
By Paul Elias, Associated Press,
http://www.usatoday.com